- 1Institute of Applied Physics, TU Wien, Vienna, Austria
- 2Institute for Planetary Research, German Aerospace Center (DLR), Berlin, Germany
- 3Laboratory for Astrophysics and Surface Physics, University of Virginia, Charlottesville, VA, USA
The development of hollows on Mercury—small depressions with spectrally bright interiors and halos—may have resulted from the selective loss of volatile components in these regions. Proposed mechanisms driving elemental or molecular volatility include intense solar radiation, micrometeoroid impacts, and repeated thermal cycling, which leave behind a refractory layer with distinct spectral properties. We investigate solar wind ions as a potential darkening agent and demonstrate that Mercury-relevant sulfides preserve their high reflectivity.
Experimental ion-irradiation studies on sulfides such as NiS, CuS, CoS, FeS, and MoS have shown that metallic surface layers can form through cation segregation and preferential sulfur depletion [1–5]. Laboratory work has further revealed iron enrichment at the surface, accompanied by visible darkening, in troilite (FeS) and more recently in pentlandite [(Fe,Ni)₉S₈] subjected to ion and laser irradiation [e.g., 6, 7]. Unlike lunar, meteoritic, and asteroidal materials, which contain significant iron and nickel sulfide minerals, Mercury’s surface appears depleted in iron. Instead, sulfides are expected to be dominated by Mn, Ti, Cr, Mg, and Ca cations, based on correlations with sulfur detected by MESSENGER’s spectrometer suite [8–10].
We have previously shown that the sulfides: niningerite (MgS) and oldhamite (CaS), two phases proposed as likely hollow-forming material based on visible-to-near-infrared (VNIR) spectral analysis [11], are radiation-hard [12]. These sulfides thereby resist the formation of a metallic top layer, observed in ion-irradiated Fe-sulfides, when exposed to solar wind-speed protons and helium ions.
Subsequent spectral analysis of the irradiated CaS and MgS showed brightening in parts of the visible range (0.4-0.7 µm) of the VNIR (Fig. 1) and an overall brightening in the mid-infrared (MIR) range observed by BepiColombo (7-14 µm, Fig. 2). The sample labeled as oxidized was exposed to air for months after the experiments, which led to an overall darkening in both VNIR and MIR, erasing the effect of irradiation. On Mercury, there is no immediate oxidation of the top layer; instead, re-deposition of thermal and ion desorbed material introduces surface contaminants, which over geological timespans could form a layer. The radiation-hard nature of sulfides could thereby act as a cleaning mechanism, where contaminants are removed continuously under solar wind exposure without damaging the bulk, preserving the reflectivity of the sample. This process provides a new framework for understanding the formation of bright hollow materials, highlighting the importance of non-transition-metal sulfides in Mercury’s surface evolution.
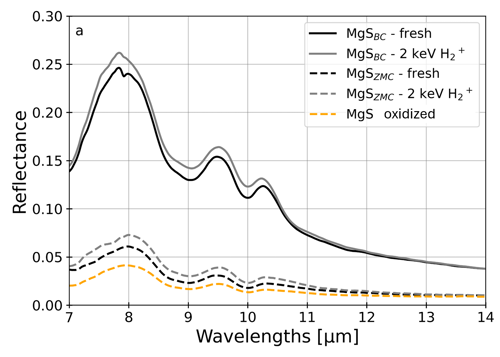
Figure 1: Visual-to-near infrared measurement of fresh MgS (black line) compared to 2 keV protons (salmon) and an old, oxidized sample (yellow). Two unirradiated MgS samples are shown, whereas one is fresh (syn) and one is old and oxidized (old).
Figure 2: Mid-infrared measurements of two MgS samples sourced from BenchChem (BC, solid lines) and Zegen Metals & Chemicals (ZMC, dashed lines). The irradiated samples (light gray) have a consistently higher reflectivity than the unirradiated (fresh) samples. The heavily oxidized sample (yellow) is the darkest.
How to cite: Jäggi, N., Barraud, O., and Dukes, C.: Self-cleaning Sulfides as Mercury Hollow Bright Material, EGU General Assembly 2026, Vienna, Austria, 3–8 May 2026, EGU26-5210, https://doi.org/10.5194/egusphere-egu26-5210, 2026.
Comments on the supplementary material
AC: Author Comment | CC: Community Comment | Report abuse
Post a comment