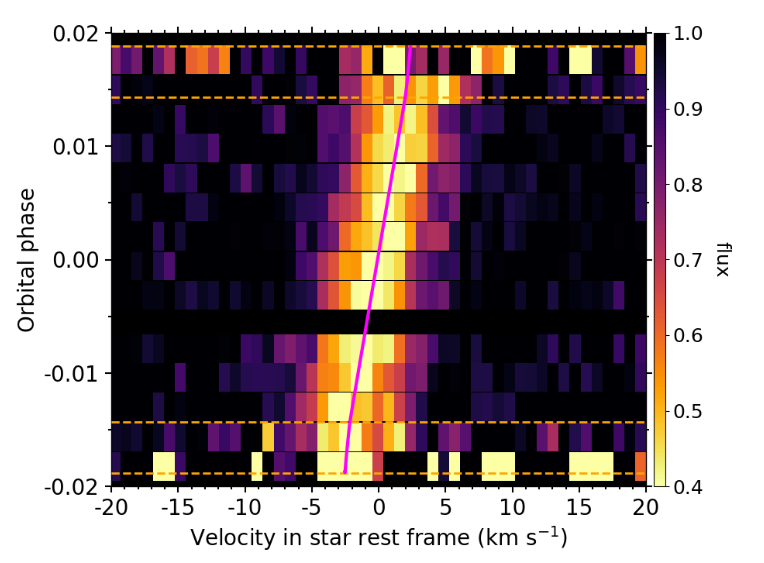
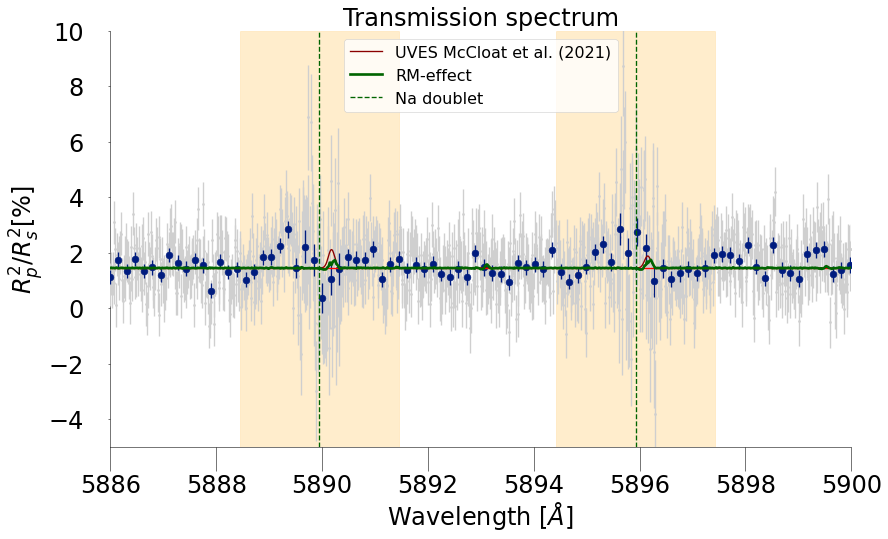
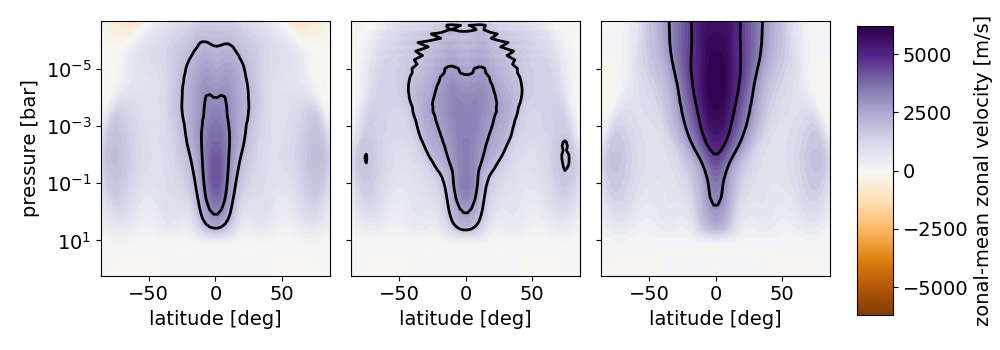
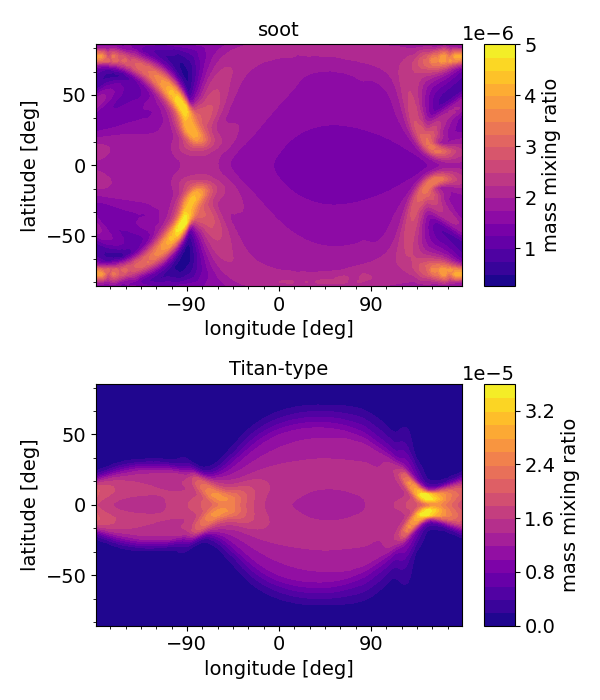
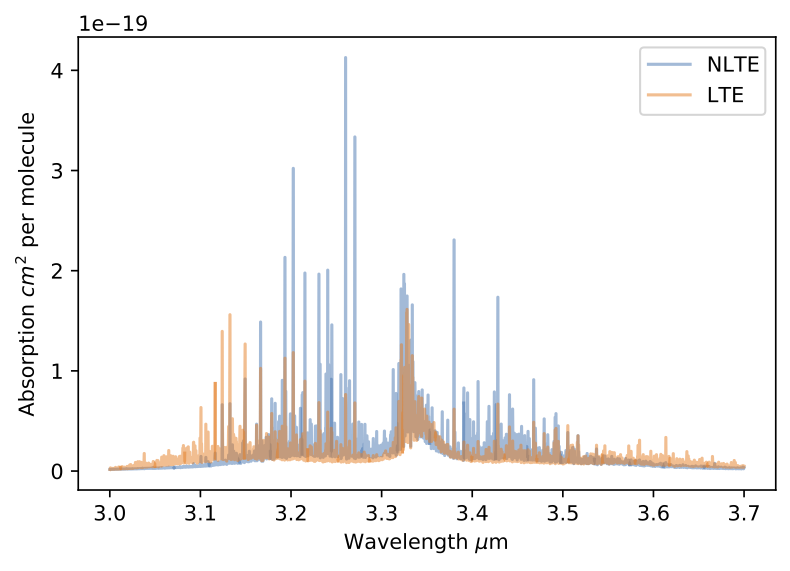
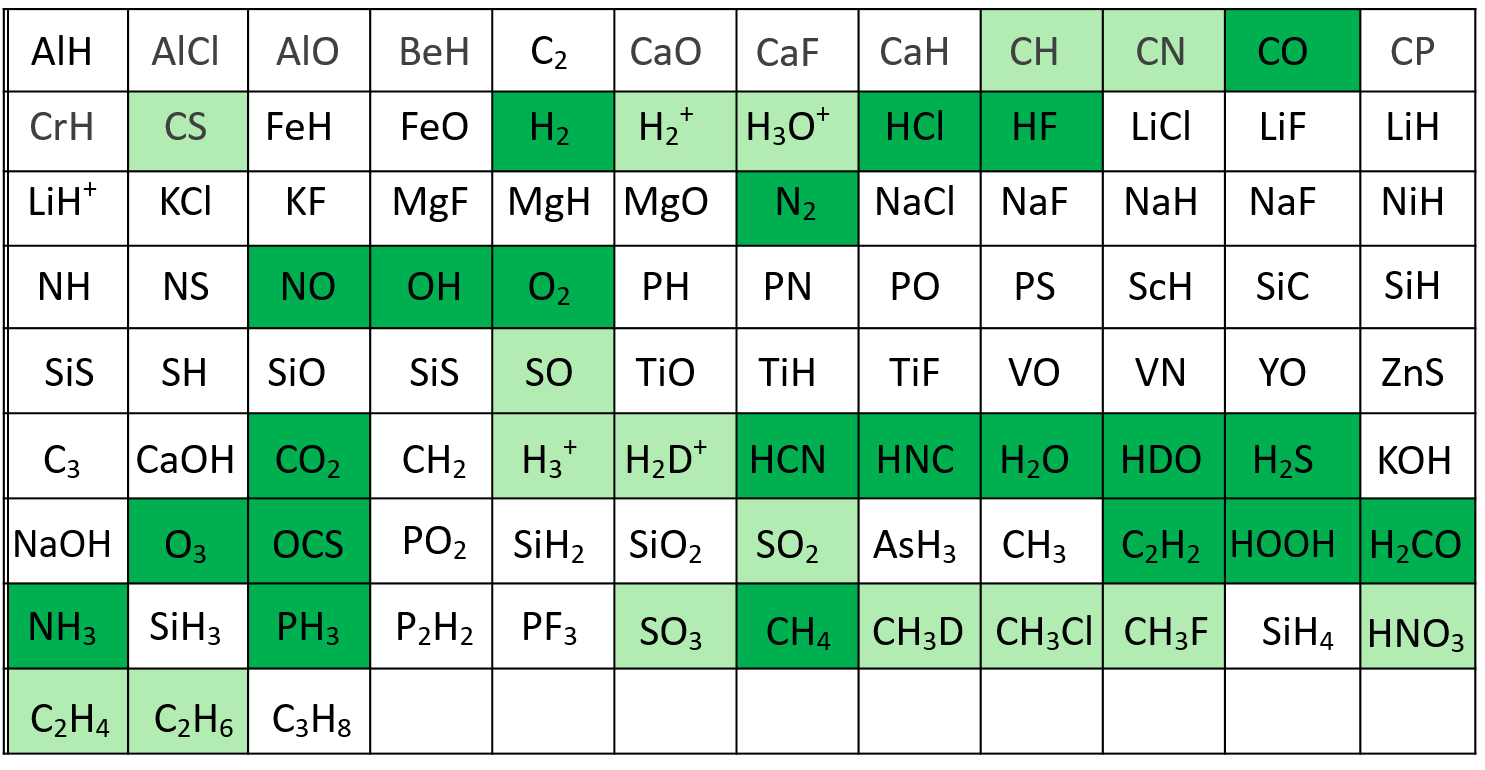
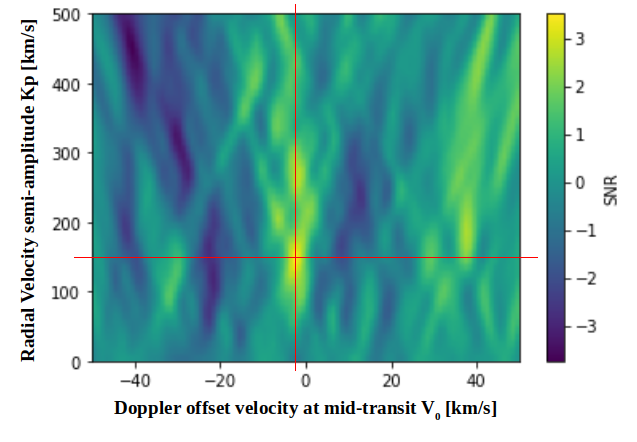
EXOA6
Exoplanet observations, modelling and experiments: Characterization of their atmospheres
Session assets
Discussion on Slack
Orals: Wed, 21 Sep, 15:30–12:10 | Room Andalucia 3
Chairperson: Monika Lendl
Terrestrial planets
15:30–15:40
|
EPSC2022-742
|
ECP
15:40–15:50
|
EPSC2022-883
15:50–16:00
|
EPSC2022-1110
|
ECP
Sub-Neptunes
16:00–16:10
|
EPSC2022-904
|
ECP
Hot Jupiters & Warm Saturns
16:20–16:30
|
EPSC2022-583
|
ECP
16:40–16:50
|
EPSC2022-133
|
ECP
Coffee break
Chairpersons: Achrène Dyrek, Mathilde Poveda
17:40–17:50
|
EPSC2022-411
|
ECP
18:00–18:10
|
EPSC2022-29
|
MI
18:10–18:20
|
EPSC2022-763
|
ECP
|
MI
Orals: Thu, 22 Sep, 12:00–17:00 | Room Albéniz+Machuca
Chairperson: Giuseppe Morello
12:00–12:10
|
EPSC2022-781
|
ECP
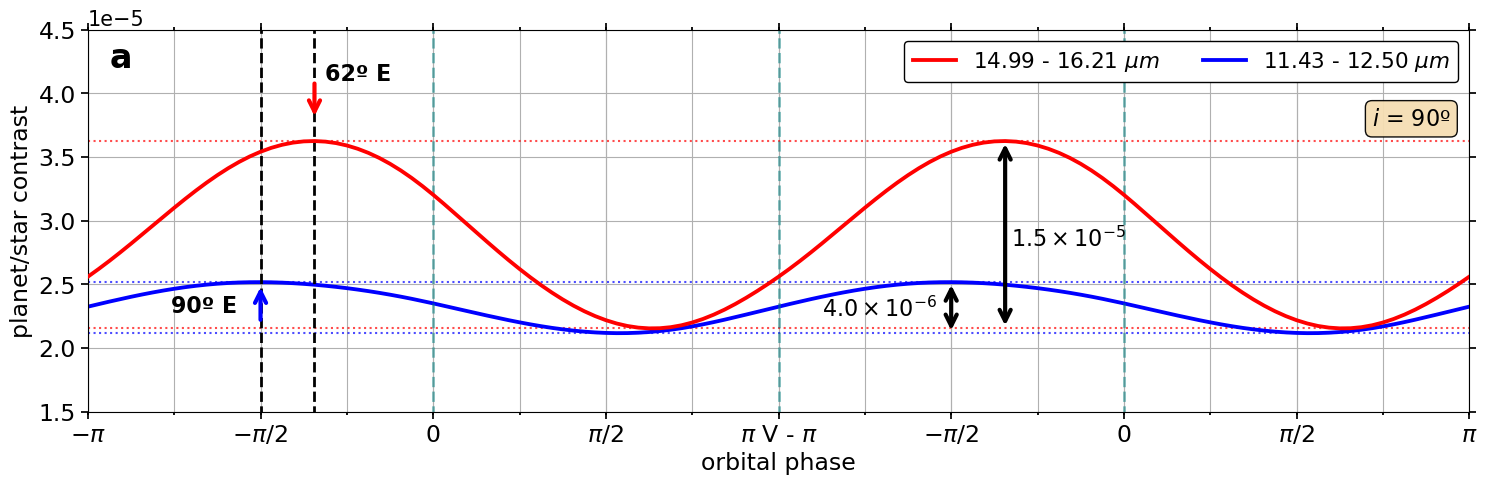
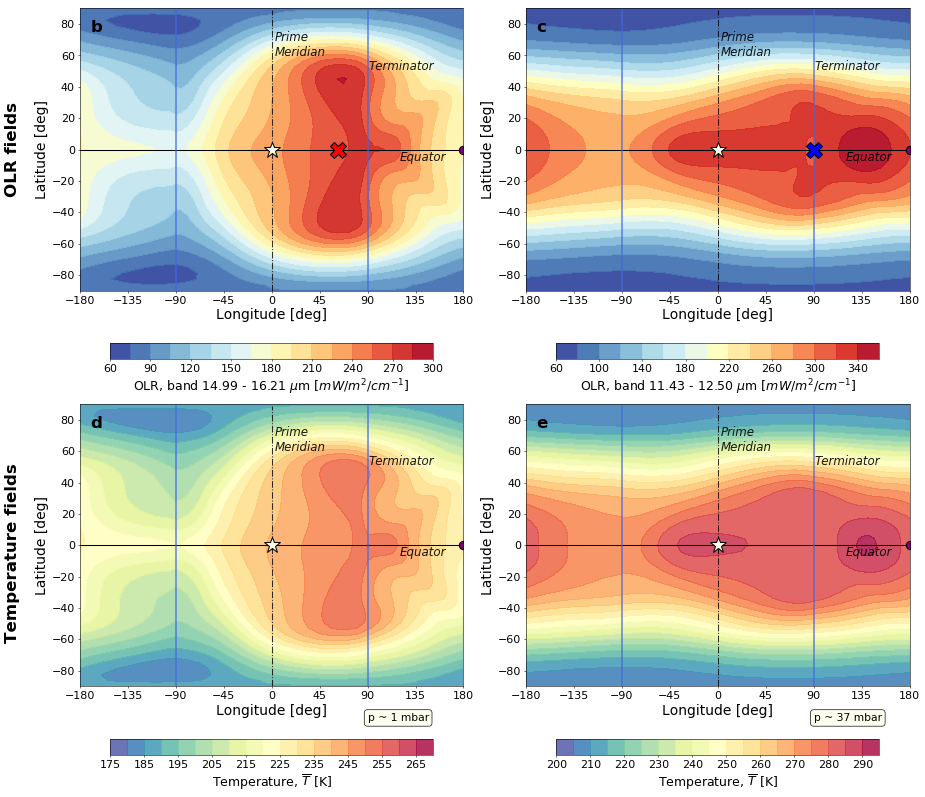
Simultaneous Phase Curve Retrieval of WASP-43b Self-consistent 3D Temperature Structure using JWST/MIRI Synthetic Observations
(withdrawn)
12:30–12:40
|
EPSC2022-1063
|
ECP
Clearing the Gridlock: Exploring Trends in Hot Jupiter Atmospheres with a Grid of 149 Parameterised Non-Grey GCM Simulations.
(withdrawn)
Ultra-hot Jupiters
12:40–12:50
|
EPSC2022-331
|
ECP
|
MI
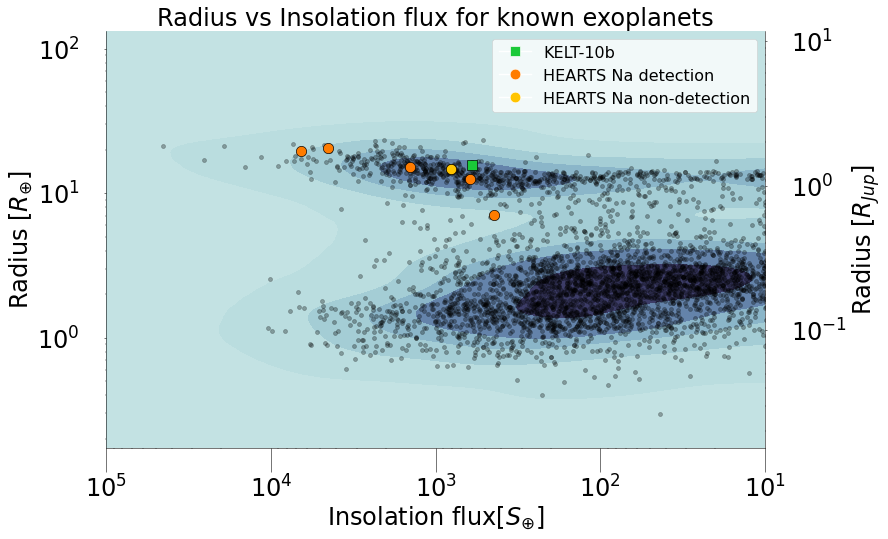
Exoplanet atmospheres in a new light: Paschen-β detection in the atmosphere of KELT-9b.
(withdrawn)
12:50–13:00
|
EPSC2022-1134
|
ECP
|
MI
13:00–13:10
|
EPSC2022-513
|
ECP
Improving techniques & Data
13:10–13:20
|
EPSC2022-1009
|
ECP
13:20–13:30
|
EPSC2022-248
|
ECP
Lunch break
Chairperson: Benjamin Fleury
16:00–16:10
|
EPSC2022-619
|
ECP
16:10–16:20
|
EPSC2022-979
|
ECP
16:20–16:30
|
EPSC2022-31
|
ECP
Towards the future telescopes
16:50–17:00
|
EPSC2022-613
|
ECP
|
MI
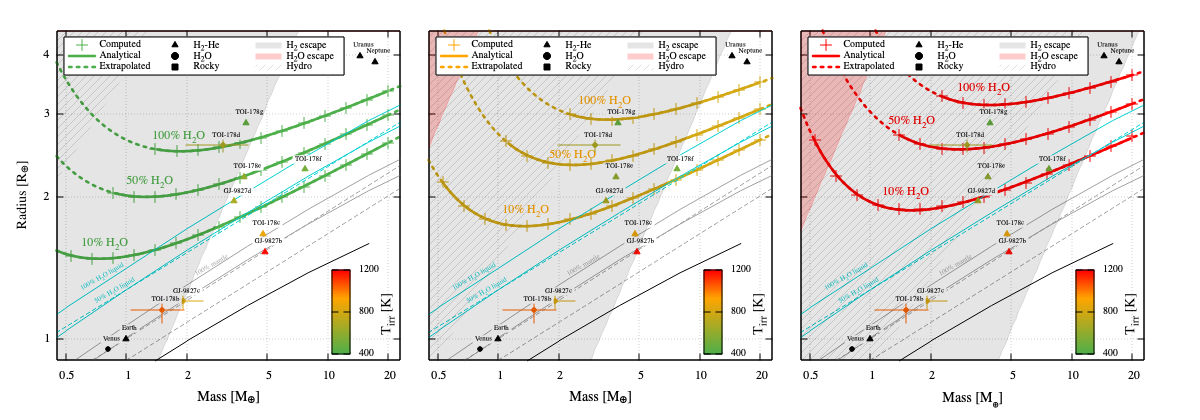
The Pursuit of A Meticulous Chemical Survey of Exoplanets
(withdrawn)
Posters: Thu, 22 Sep, 18:45–20:15 | Poster area Level 2
L2.38
|
EPSC2022-1181
|
ECP
L2.39
|
EPSC2022-1178
ExoAtmospheres: a community database for exoplanet atmospheres research
(withdrawn)